| 1. |
A particle with a +2 charge which is produced during the decay of a radioactive element is:
|
|
|
|
|
|
a)
|
a proton.
|
|
b)
|
a deuteron.
|
|
c)
|
a positron.
|
|
d)
|
an alpha particle
|
|
e)
|
a beta particle.
|
|
|
|
|
|
|
| 2. |
An atom with 15 protons and 18 neutrons would be an isotope of:
|
|
|
|
|
|
a)
|
arsenic
|
|
b)
|
phosphorus
|
|
c)
|
argon
|
|
d)
|
sulfur
|
|
e)
|
chlorine
|
|
|
|
|
|
|
| 3. |
What is the mass defect for  ? ?
Masses:  = 11.0216; proton = 1.0073; neutron =
1.0087; electron = 0.00055 amu. = 11.0216; proton = 1.0073; neutron =
1.0087; electron = 0.00055 amu.
|
|
|
|
|
|
a)
|
1.685 amu
|
|
b)
|
0.1707 amu
|
|
c)
|
0.1055 amu
|
|
d)
|
0.0707 amu
|
|
e)
|
0.0055 amu
|
|
|
|
|
|
|
| 4. |
A positron has the same mass as:
|
|
|
|
|
|
a)
|
a gamma ray.
|
|
b)
|
a neutron.
|
|
c)
|
an alpha particle.
|
|
d)
|
a proton.
|
|
e)
|
a beta particle.
|
|
|
|
|
|
|
| 5. |
 can
be prepared by electron capture from: can
be prepared by electron capture from:
|
|
|
|
|
|
a)
|
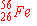
|
|
b)
|
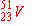
|
|
c)
|
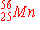
|
|
d)
|
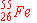
|
|
e)
|
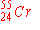
|
|
|
|
|
|
|
| 6. |
In the ejection of a beta particle from the nucleus, the following occurs.
|
|
|
|
|
|
a)
|
A positron is converted to a neutron.
|
|
b)
|
A neutron is converted to a positron.
|
|
c)
|
A neutron is converted to a proton.
|
|
d)
|
A proton is converted to a neutron.
|
|
e)
|
A neutron is converted into an electron.
|
|
|
|
|
|
|
| 7. |
Among the following nuclides, the highest nuclear binding energy per nucleon is found for:
|
|
|
|
|
|
a)
|
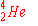
|
|
b)
|
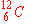
|
|
c)
|
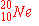
|
|
d)
|
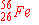
|
|
e)
|
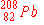
|
|
|
|
|
|
|
| 8. |
The most likely mode of decay for 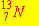 is: is:
|
|
|
|
|
|
a)
|
alpha emission.
|
|
b)
|
beta emission.
|
|
c)
|
positron emission.
|
|
d)
|
gamma radiation.
|
|
e)
|
a neutron emission.
|
|
|
|
|
|
|
| 9. |
Of the following nuclides, the one most likely to be radioactive is:
|
|
|
|
|
|
a)
|
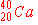
|
|
b)
|
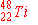
|
|
c)
|
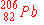
|
|
d)
|
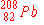
|
|
e)
|
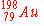
|
|
|
|
|
|
|
| 10. |
The half-life of the radioisotope  is 1.0 hr. The decay of a 160-g sample of
the isotope to 1.25 grams requires: is 1.0 hr. The decay of a 160-g sample of
the isotope to 1.25 grams requires:
|
|
|
|
|
|
a)
|
3.0 hr.
|
|
b)
|
4.0 hr.
|
|
c)
|
5.0 hr.
|
|
d)
|
6.0 hr.
|
|
e)
|
7.0 hr.
|
|
|
|
|
|
|
| 11. |
The nuclear bombardment reaction
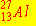 + +   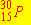 + + 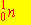
is represented by:
|
|
|
|
|
|
a)
|
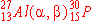
|
|
b)
|
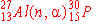
|
|
c)
|
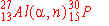
|
|
d)
|
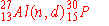
|
|
e)
|
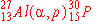
|
|
|
|
|
|
|
| 12. |
The most probable mode of decay for  is: is:
|
|
|
|
|
|
a)
|
alpha emission.
|
|
b)
|
beta emission.
|
|
c)
|
positron emission.
|
|
d)
|
neutron emission.
|
|
e)
|
gamma emission.
|
|
|
|
|
|
|
| 13. |
When  undergoes an electron capture, what is the
immediate nuclear product? undergoes an electron capture, what is the
immediate nuclear product?
|
|
|
|
|
|
a)
|
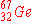
|
|
b)
|
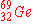
|
|
c)
|
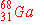
|
|
d)
|
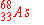
|
|
e)
|
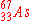
|
|
|
|
|
|
|
| 14. |
A living tree contains  (half-life 5600 years) and has a specific activity
of 750 counts per hour. A wooden artifact from archeological site gives a count of 90
counts per hour. The age of this artifact is most nearly: (half-life 5600 years) and has a specific activity
of 750 counts per hour. A wooden artifact from archeological site gives a count of 90
counts per hour. The age of this artifact is most nearly:
|
|
|
|
|
|
a)
|
5600 years.
|
|
b)
|
11,000 years.
|
|
c)
|
17,000 years.
|
|
d)
|
22,000 years.
|
|
e)
|
47,000 years.
|
|
|
|
|
|
|
| 15. |
When  undergoes neutron emission, what is the immediate
nuclear product? undergoes neutron emission, what is the immediate
nuclear product?
|
|
|
|
|
|
a)
|
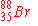
|
|
b)
|
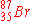
|
|
c)
|
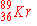
|
|
d)
|
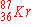
|
|
e)
|
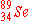
|
|
|
|
|
|
|
| 16. |
When  undergoes positron emission, what is the
immediate nuclear product? undergoes positron emission, what is the
immediate nuclear product?
|
|
|
|
|
|
a)
|
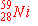
|
|
b)
|
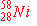
|
|
c)
|
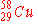
|
|
d)
|
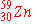
|
|
e)
|
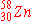
|
|
|
|
|
|
|
| 17. |
As a radioactive isotope decays, its half-life:
|
|
|
|
|
|
a)
|
doubles.
|
|
b)
|
halves.
|
|
c)
|
decreases.
|
|
d)
|
increases.
|
|
e)
|
remains the same.
|
|
|
|
|
|
|
| 18. |
The isotope 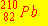 has a half-life of 22 years. What percentage of
a pure has a half-life of 22 years. What percentage of
a pure 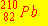 sample prepared in April 1937 remains in April 1993? sample prepared in April 1937 remains in April 1993?
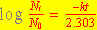 and
and 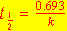
|
|
|
|
|
|
a)
|
38%
|
|
b)
|
31%
|
|
c)
|
26%
|
|
d)
|
21%
|
|
e)
|
17%
|
|
|
|
|
|
|
| 19. |
What is the decay constant for iodine-131, which is used to traet cancer of the thyroid. It decays by beta emission with a half-life of 8.05 days.
|
|
|
|
|
|
a)
|
8.35 x 10-2 day-1
|
|
b)
|
1.59 x 10-3 hr-1
|
|
c)
|
3.97 x 103 hr-1
|
|
d)
|
5.98 x 10-5 min-1
|
|
e)
|
1.43 x 10-6 sec-1
|
|
|
|
|
|
|
| 20. |
A 4.50-mg sample of a newly discovered isotope was analyzed and found to contain only 3.25 mg after a period of 29.6 hours. What is the half-life of the isotope?
|
|
|
|
|
|
a)
|
47.6 hr
|
|
b)
|
53.4 hr
|
|
c)
|
58.0 hr
|
|
d)
|
63.0 hr
|
|
e)
|
71.2 hr
|
|
|
|
|
|
|
| 21. |
Strontium-90 is produced in nuclear explosions. It can replace calcium in the bones. The half-life of Sr-90 is 27.7 years. What is the rate constant for the decay of Sr-90?
|
|
|
|
|
|
a)
|
0.0125 yr-1
|
|
b)
|
0.0250 yr-1
|
|
c)
|
0.0375 yr-1
|
|
d)
|
0.0425 yr-1
|
|
e)
|
0.0450 yr-1
|
|
|
|
|
|
|
| 22. |
If the activity of Strontium-90 in the bones of an exposed person was 90 dps, how long will it take the activity (in disintegrations per second) to decrease to 3.5 dps?
|
|
|
|
|
|
a)
|
50 yr
|
|
b)
|
65 yr
|
|
c)
|
70yr
|
|
d)
|
95 yr
|
|
e)
|
130 yr
|
|
|
|
|
|
|
| 23. |
What is the rate constant of decay for Cobalt-60, which has a ha;f-life of 5.27 years?
|
|
|
|
|
|
a)
|
0.113 yr
|
|
b)
|
0.161 yr
|
|
c)
|
3.59 x 10-4 day-1
|
|
d)
|
7.18 x 10-3 hr-1
|
|
e)
|
25.8 sec-1
|
|
|
|
|
|
|
| 24. |
The half-life of Phosphorus-33 is 25 days. How much of a 128-g sample will remain after 150 days?
|
|
|
|
|
|
a)
|
16 g
|
|
b)
|
8 g
|
|
c)
|
4 g
|
|
d)
|
2 g
|
|
e)
|
1 g
|
|
|
|
|
|
|
| 25. |
What is the binding energy per nucleon, in joules, of a chlorine-35 atom, given that the atomic mass equals 34.9595 amu?
(e-1 mass = 0.00055 amu; n mass = 1.00867 amu; p mass = 1.00728 amu;
c = 3.00 x 108 m/s; 1 amu = 1.6606 x 10-24 g.)
|
|
|
|
|
|
a)
|
1.41 x 10-12 J/nucleon
|
|
b)
|
4.93 x 10-11 J/nucleon
|
|
c)
|
1.56 x 10-12 J/nucleon
|
|
d)
|
1.56 x 10-11 J/nucleon
|
|
e)
|
1.56 x 10-10 J/nucleon
|
|
|
|
|
|
|